Definition: A secondary cell can be recharged or restored. The chemical reaction that occurs on discharge may be reversed by forcing a current through the battery in the opposite direction. This charging current must be supplied from another source, which can be a generator or a power supply.
Figure 1 shows one type of battery charger used for recharging automobile and motorcycle batteries. An alternating current, which will be studied in a later chapter, must be rectified to a direct current for charging the battery.
Figure 1. This type of charger is called a trickle charger. It slowly brings a battery back to full charge.
Lead Acid Cell
A common type of lead acid cell is the car storage battery. A storage battery does not store electricity. Rather, it stores chemical energy, which in turn produces electrical energy.
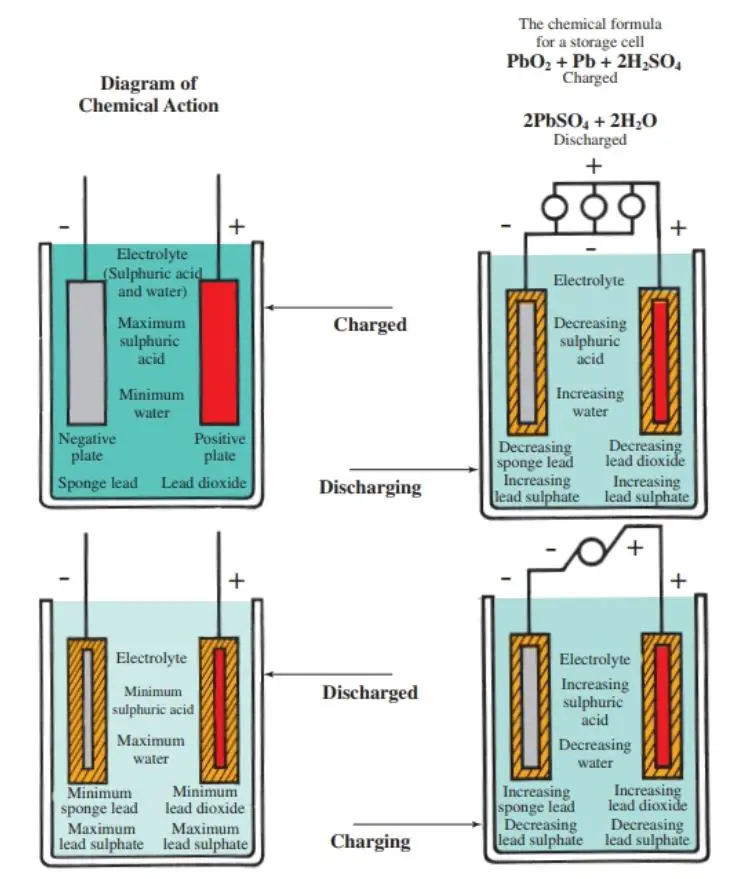
The active ingredients in a fully charged battery are lead peroxide (PbO2), which acts as the positive plate, and pure spongy lead (Pb) for the negative plate.
The liquid electrolyte is sulfuric acid (H2SO4) and water (H2O). The positive plates are a reddish-brown color. Negative plates are gray.
The chemical reaction is rather involved. However, study the information given in Figure 2. Notice that during discharge, both the spongy lead and the lead peroxide (also called lead dioxide) plates are being changed to lead sulfate, and the electrolyte is being changed to water.
When the cell is recharged, the reverse action occurs. The lead sulfate changes back to spongy lead and lead peroxide; the electrolyte to sulfuric acid.
Figure 2. How a lead-acid cell works. (ESB Brands, Inc.)
The electrolyte of a fully charged battery is a solution of sulfuric acid and water. The weight of a pure sulfuric acid is 1.835 times heavier than water. This is called its specific gravity.
A Lesson in Safety:
During the charging process of a storage battery, highly explosive hydrogen gas may be present. Do not smoke or light matches near charging batteries. Charge only in a well-ventilated room.
Batteries should be first connected to the charger before the power is applied. Otherwise, the sparks made during connection might ignite the hydrogen gas and cause an explosion.
Specific gravity is the weight of a liquid as it compares to water. The specific gravity of water is 1.000. The acid and water mixture in a fully charged battery has a specific gravity of approximately 1.300 or less.
As the electrolyte changes to water when the cell discharges, the specific gravity becomes approximately 1.100 to 1.150. Therefore, the specific gravity of the electrolyte can be used to determine the state of charge of a cell.
The instrument used to measure the specific gravity is a hydrometer. The principle of the hydrometer is based on Archimedes principle in physics.
This principle states that a floating body will displace an amount of liquid equal to its own weight.
If the cell is in a fully charged state, the electrolyte liquid is heavier, so the float in the hydrometer will not sink as far. The distance that the float does sink is calibrated in specific gravity on the scale. This can be read as the state of charge of the cell.
Caution
Expensive storage batteries may be destroyed by excessive vibration and rough handling. Chemicals may break off from the plates and cause internal short circuits and dead cells. Handle a battery gently and be sure it is securely clamped and bolted in your car.
In the 12-volt automotive battery, six lead-acid cells are placed in a molded hard rubber case. Each cell has its own compartment.
At the bottom of each compartment space, or sediment chamber is provided. This is where particles of chemicals broken from the plates due to chemical action or vibration can collect. Otherwise, these particles would short out the plates and make a dead cell. The individual cells are connected in series by lead alloy connectors.
Caution
Automotive batteries contain large amounts of lead. Consequently, they should never be disposed of in landfills. Stores that sell automotive batteries are required by law to accept old batteries for recycling.
Nickel-Cadmium cell
The nickel-cadmium cell is a rechargeable dry cell. Basically, these are nickel-cadmium alkaline batteries with paste rather than liquid for the electrolyte.
The ability to be recharged is just one of their advantages. Other advantages include long life, high efficiency, compactness, and lightweight.
The nickel-cadmium cell produces a high discharge current due to its low internal resistance. Other uses include the powering of small radios, burglar alarm systems, camera flashes, and aircraft instruments.
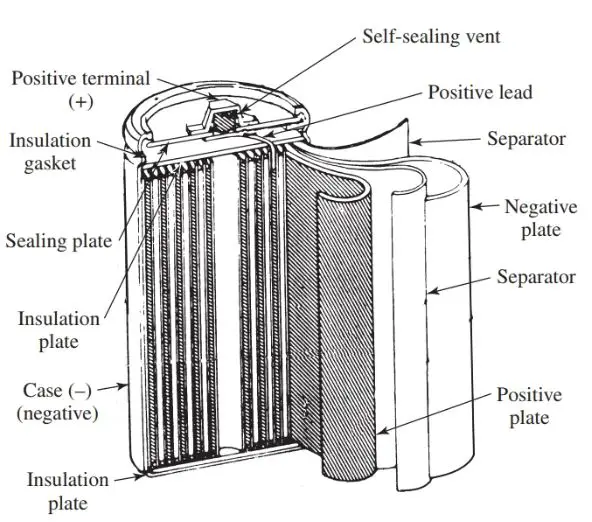
One type of nickel-cadmium cell uses positive and negative plates, a separator, alkaline electrolyte, a metal case, and a sealing plate with a self-resealing safety vent. It is shown in Figure 3.
The positive plate of this battery is a porous, powdered nickel base plate. It is filled with nickel hydroxide.
The negative plate is a punched plate of thin steel, coated with cadmium active material. The separator is made of a polyamide fiber. For high-temperature uses, it is made of a nonwoven polypropylene fiber.
The positive plate, separator, and negative plate are pressed together, wound into a coil, and inserted in the metal case.
Figure 3. Construction of a nickel-cadmium cell. (Panasonic Battery Sales Division)
The electrolyte is an alkaline aqueous solution. It is totally absorbed into the plate and separator.
The metal case is constructed of nickel-plated steel. It is welded on the inside to the negative plate. It becomes the negative pole.
The sealing plate uses a special liquid sealing agent to form a perfect seal. The positive plate is welded on the inside to the sealing plate. It becomes the positive pole.
The self-resealing safety vent permits the discharge of gas in the event of an abnormal increase of internal pressure. This prevents against rupture or other damage.
The vent is made of a special alkaline and oxidation resistant rubber. This ensures that operating pressure and safety features will be retained over a long period of time.
The electromechanical processes of a nickel-cadmium alkaline cell are outlined below.
\[\begin{align} & \begin{matrix} 2NiOOH & + & \begin{matrix} Cd & + & \begin{matrix} 2{{H}_{2}}O & \leftrightarrow & \begin{matrix} 2Ni{{\left( OH \right)}_{2}} & \begin{matrix} + & Cd{{\left( OH \right)}_{2}} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\ & \begin{matrix} Positive & {} & \begin{matrix} Negative & Electrolyte & \begin{matrix} Positive & {} & \begin{matrix} Negative & \begin{matrix} {} & {} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\\end{matrix} \\\end{align}\]
In this process, charging and discharging are reversed in a very efficient manner. The electrical energy used during discharge is regained during recharge. During the final charging stage, an oxygen gas is created with the reaction occurring at the positive.
\[\begin{align} & \begin{matrix} \begin{matrix} \begin{matrix} 4O{{H}^{-}} & \to & {{O}_{2}}\uparrow \\\end{matrix} & + & 2{{H}_{2}}O & + \\\end{matrix} & 4{{e}^{-}} & {} & {} \\\end{matrix} \\ & \begin{matrix} Hydroxide & Oxygen & Water & Electrons \\\end{matrix} \\\end{align}\]
This oxygen passes through the separator to the negative. After this, an absorption reaction takes place at the negative and absorption occurs.
\[\begin{align} & \begin{matrix} \begin{matrix} \begin{matrix} {{O}_{2}} & + & 2{{H}_{2}}O \\\end{matrix} & + & 4{{e}^{-}} & \to \\\end{matrix} & 4O{{H}^{-}} & {} & {} \\\end{matrix} \\ & \begin{matrix} Oxygen & Water & Electrons & Hydroxide-Ions \\\end{matrix} \\\end{align}\]
Nickel Metal Hydride Cell
Nickel metal hydride (NiMH) is a rechargeable battery very similar to the NiCad battery.
The NiMH battery uses nickel for the positive cathode and a hydrogen-absorbing alloy for the negative anode rather than cadmium, which is found in the NiCad battery.
The fact that NiMH batteries do not use cadmium makes them environmentally friendly.
Hydrogen-absorbing alloys are made from combinations of nickel and iron (Fe), magnesium (Mg), and lanthanum (La). The alloys can absorb large volumes of hydrogen equal to approximately 1000 times their weight without an increase in volume. This means the batteries can be sealed without the danger of explosion caused by the expansion of materials.
A NiMH battery can have two to three times the storage capacity of a NiCad battery of equal size. This has made it an excellent battery for hybrid vehicles and portable computer applications.
The discharge voltage level is 1.2 volts, which is the same as for NiCad batteries. The NiMH battery can be recharged over 500 times. The combined characteristics of the NiMH makes it an ideal replacement for existing NiCad battery applications.
NiMH batteries should never be completely discharged. If they are, one or more of the cells can reverse polarity, which will result in permanent damage to the battery.
Electronic devices that use NiMH batteries typically have a special circuit designed to detect low voltage levels and then disconnect the battery circuit before damage can occur.