The article discusses electrolytic corrosion, particularly the issues arising from contact between dissimilar metals in the presence of moisture and electrolytes. It also covers methods of preventing such corrosion, including insulation, sacrificial anodes, cathodic protection, and minimizing exposure to stray currents.
Electrolytic Corrosion
Electrolytic corrosion is created by contact between two different metals in a damp atmosphere with dust or pollution of a type that will dissolve in water. Precautions may have to be taken to ensure that the join between two different metals is kept dry.
Dissimilar Metals
The best-known effect of destructive corrosion occurs when two different metals are in contact in the presence of an electrolyte.
An increasing problem in Australia is the connection of copper conductors to aluminum conductors. The copper and aluminum act as electrodes when an electrolyte is present from condensation or rain that contains dissolved sulfur products. A simple cell is created and is effectively short-circuited on itself, as illustrated in Figure 1.

Figure 1 Anodized Aluminum Torch
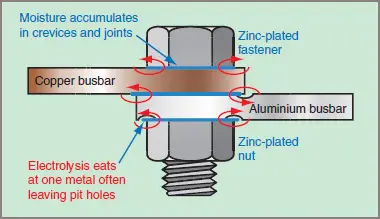
Figure 2 Corrosion of Dissimilar Metals
The metal with the higher potential becomes the anode that will tend to go into solution in the electrolyte (i.e. will corrode away). The result is a progressively deteriorating electrical connection, the generation of heat and the eventual failure of the connection. In some circumstances, the heat produced can set fire to an installation and the problem is then more than just the failure of an electric circuit.
One way to combat this type of corrosion is to minimize the number of connections where dissimilar metals are joined. Another method is to prevent the entry of moisture to the joint by applying paint or other covering. Alternatively, all the materials being joined can be electroplated with a common metal, for example, cadmium.
Stray Electric Currents
Underground pipes and cables can become corroded by the electrolytic action of stray underground currents. Dissimilar metals in damp ground and adjacent to each other may produce the stray currents or the currents may result from a nearby faulty electrical installation.
The electrical bonding of tracks in electric traction systems helps to reduce underground stray currents, and the covering of underground pipes and conductors with plastic sheaths minimizes the corrosive effects. The corrosion tends to be concentrated at sharp bends in underground pipework and appropriate measures to avoid this should be taken where practicable.
Electrolytic Protection — Sacrificial Anodes
Electrolytic corrosion can be minimized by implementing sacrificial anodes. Anodes made of zinc blocks are bolted inside boilers or to the metal hulls of ships, adjacent to bronze propellers. The intention is to sacrifice a metal block, which can be replaced comparatively easily, rather than risk the corrosion of the boiler or ship’s hull.
Electrolytic Protection — Cathodic Protection
With this method, an external DC voltage is applied between the equipment to be protected and earth, such that the equipment is at a lower potential than the surrounding soil. In practice, it has been found that most forms of corrosion can be prevented when the pipes or other metallic structures are approximately 0.5 V negative with respect to the surrounding soil.
Electrolytic Corrosion Key Takeaways
Understanding and preventing electrolytic corrosion is essential for ensuring the longevity and safety of electrical systems, underground infrastructure, and marine equipment. By applying protective measures such as insulation, sacrificial anodes, and cathodic protection, industries can avoid costly damage, maintain efficient operation, and reduce the risk of system failures or hazards, making these practices crucial in real-world applications.