In order to compare batteries, an electrician must first know what parameters (specifications) to consider.
Terminal Voltage
The most identifiable measure of a cell is the ‘terminal voltage’, which at first may seem too obvious to be so simple. In fact, the terminal voltage can change dramatically as a cell goes through charge and discharge cycles. The ‘nominal voltage’ is what the chemists tell us the cell should produce with zero current flowing.
Whenever a current is drawn from a cell or pushed into a cell, the voltage changes, even when the current is that drawn by a voltmeter. The reason is that any real cell has a real resistance within the cell, known as the ‘internal resistance’ (see Figure 1).
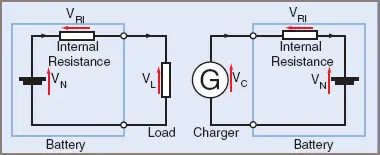
Figure 1 Internal Resistance
The internal resistance will have a voltage drop across it that adds to the nominal voltage during the charging cycle and subtracts from the nominal voltage during the discharge cycle.
![]()
Figure 2 shows the effects of internal resistance on terminal voltage for various load currents.
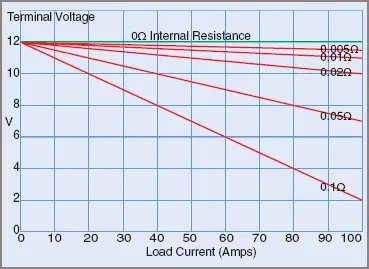
Figure 2 Battery Terminal Voltage Drop
Energy Capacity
The energy that a cell can store depends on the chemistry and the physical size of the plates, mostly the area, but to some extent the thickness of the plates for some chemistries.
Ideally, the energy storage should be measured in joules, mega joules for sufficiently large battery banks. However, convention has us working in ampere-hours (Ah), the number of amps a battery can deliver in a certain number of hours.
The problem here is that ampere-hours do not take into account the voltage of the battery and so two batteries of the same physical size may have a different number of cells, and therefore a different ampere-hour rating, even though the energy stored may be the exact same quantity in mega joules.
A battery with a stated capacity of 10 Ah can in simple terms provide 10 amps for 1 hour or 1 amp for 10 hours. However, this does not take into account the internal resistance of the battery, which changes with the condition of the battery.
In 1897 a German physicist, W. Peukert, determined that the capacity of a lead–acid battery depends on the discharge rate of the battery, saying that high discharge rates decrease the storage capacity by a predictable factor.
\[{{C}_{P}}={{I}^{k}}t\]
Where:
- C is the capacity in Ah @ 1 amp discharge.
- I is the actual discharge current in amps.
- k is the Peukerts constant for the battery.
- t is the discharge time in hours.
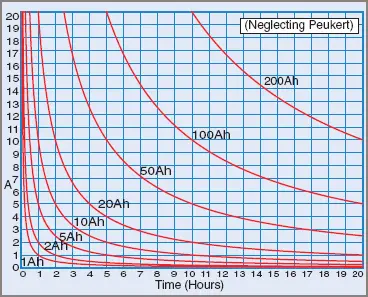
Figure 3 Battery Ampere Capacity
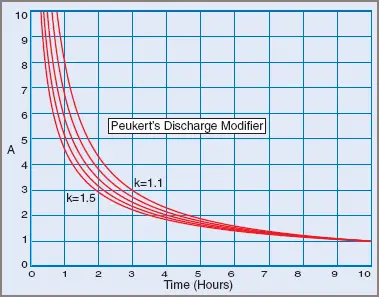
Figure 4 Peukert’s discharge modifier
This means that, for a typical 10 Ah battery with a Peukert constant of 1.2, a 10 A discharge rate will discharge the battery in just 0.63 hours or 63 per cent of the expected time.
Note that Peukert’s equation holds true for other types of cell technology, but the Peukert’s constant must be known for the cell type and age.
Specific Gravity (SG)
When acid is mixed with water, the specific gravity of the resulting electrolyte will be between that of water, which is 1 kg per liter or an SG of 1.000, and that of sulphuric acid which, at 100 per cent pure, has an SG of 1.84 or 1.84 kg per liter.
When mixed ready for use in a lead–acid battery, the SG of the diluted sulphuric acid (battery acid) is 1.250 or 1.25 kg per liter. As the battery is charged or discharged, the proportion of acid in the electrolyte changes, so the SG also changes, according to the state of charge of the battery.

Figure 5 SG test of an automobile battery
State Of Charge (SOC)
The state of charge of a battery can often be determined from the condition of the electrolyte. In a lead–acid battery, for example, the specific gravity of the electrolyte indicates the state of charge of the battery.
Other batteries may indicate the SOC by the terminal voltage.
Depth of Discharge (DoD)
Of more concern in storage batteries is the depth of discharge, as some batteries will fail if allowed to become completely flat, while others will be severely aged by deep discharge cycles.
Automotive batteries, for example, are not expected to be deeply discharged and are rapidly degraded by excessive and repetitive deep discharges. Batteries intended for electric vehicles are designed to be more tolerant of deep discharges.
Temperature
Some cell technologies are more affected by temperature than others; with some, cell temperature can be critical, with some batteries freezing at just below 0°C, while others will not operate below 200°C. Therefore the correct operating range of a battery should be known before taking it to either extreme.
Specific Energy (SE)
The specific energy of a battery refers to the energy which that type of cell can store per kilogram.
Typical figures (MJ/kg) are:
| Leclanché-type zinc–carbon cell | 0.072 0.144 MJ/kg |
| sealed zinc–mercury cell | 0.324–0.396 MJ/kg |
| silver oxide–cadmium cell | 0.144–0.216 MJ/kg |
| silver–zinc cell | 0.324–0.540 MJ/kg |
| zinc–alkaline manganese cell | 0.216–0.252 MJ/kg |
| lead–acid cell | 0.120–0.160 MJ/kg |
Specific Volume (SV)
Specific volume, on the other hand, is the energy stored per liter of volume or, to put it another way, the energy per cubic decimeter of space. Again using a lead–acid battery example, the SV might be 0.331 MJ/L. By comparison, a lithium–manganese battery is six times smaller with an SV of ~2 MJ/L.
Cold Cranking Amps
In automotive terms, the maximum current expected from a battery is called the Cold Cranking Amps, or CCA, which defines the current available to turn an engine over in cold conditions. The term may be used in other applications as well, being a leftover from the more common automotive use of batteries. The CCA rating is then the maximum short-term current draw from a battery.
Efficiency (Discharge/Charge) %
The efficiency of a battery, as with anything, is output/input × 100%. A lead–acid battery at first had an efficiency of about 75%, but thankfully has improved with efficiencies to around 95% with some technologies.
Final Voltage
The term ‘final voltage’ designates the minimum useful and accepted voltage of a cell or battery at various rates of discharge.
Cycle Life
Batteries have an inherent limitation as to the number of times they can be discharged and recharged, and you have seen that this can be reduced by excessive temperatures and depth of discharge. However, some modern technologies are now promoting 10 000 cycles of lifespan, whereas most current technologies are proud to reach 1000 cycles, with NiCd reaching 2000 cycles, under perfect conditions of course.