The article outlines essential battery maintenance practices to ensure longevity and safe operation, including regular inspections, proper charging methods, and environmental considerations. It also discusses advanced charging techniques and the use of battery management systems, particularly in modern and high-value battery applications.
A maintenance routine is recommended in order to achieve maximum service life from batteries. Checklist to observe is as follows:
| 1. | Ensure that all terminal lugs are clean and tight, to give good electrical contact. |
| 2. | Ensure that connecting straps and cables are of a suitable material and in good condition. The electrode potential of the two materials should be as close as possible. |
| 3. | For wet cells, check the electrolyte level regularly and keep it within the specified levels. The plates must be kept covered so they are not allowed to dry out.
With lead–acid cells in particular, drying out leads to a reduction in ampere-hour capacity. Unless specified otherwise, approximately 3 to 4 mm of electrolyte above the plates is usually sufficient. Some types of battery construction have been designed to reduce electrolyte loss. Special filler caps have been fitted to allow the cell to release the hydrogen and oxygen gases generated while retaining vapors. The escaping gases result from the splitting of water molecules, which leads to a reduction in electrolyte level, which must be topped up with demineralized water, otherwise known as distilled water. |
| 4. | The battery case must be kept clean of electrolyte, to prevent leakage current across the external case. Leaking acid is toxic, corrosive and may hide the extent of cracks. |
| 5. | The battery must be suitably protected from extreme temperatures and positioned where it is less likely to be physically damaged. |
| 6. | The battery must be kept in a ventilated position and not be charged or discharged at excessively high current rates, which cause excessive heat and gassing. |
| 7. | The rates of current charge or discharge must be controlled within specified design levels and time limits. |
| 8. | Wet cells should be measured for terminal voltages and the specific gravity of the electrolyte. Any discrepancy greater than 5 per cent, or as specified by the manufacturer, should be recorded and reported. Cells should all have the same readings. |
Voltage On Load
Cells that are getting old typically have an increasing internal resistance, which means that the terminal voltage goes lower under load and higher under charge.
One test of a battery or, more typically, a cell’s condition is to test the battery or cell under load. The load is a resistive load designed to draw half the rated maximum current from the battery for 15 seconds. The battery voltage should not drop below a value given on the manufacturer’s table, typically 9.5 volts for a 12-volt lead–acid battery.
Charge Rates
Batteries should be charged according to their type and the manufacturer’s specifications. The type of charge varies from type to type, with some cells given a fast charge or boost charge, while others require a specially controlled constant-voltage or constant-current charge.
Once fully charged, some batteries require a trickle charge, which is intended to overcome the effect of self-discharge. Left off charge, some batteries will go flat in a very short time, as the self-discharge rate of some batteries rises rapidly with temperature.
Modern battery chargers can be much more complex than older technology, with computer-managed charging now used. Computers monitor the battery’s voltage and charge current, and even test the battery’s internal resistance, then charges it with the most suitable method for the condition of the battery. Some systems may even monitor the electrolyte temperature, level and pH.
As shown in Figure 1, computerized chargers may begin with a constant-current charge until a certain voltage is reached, then boost charge by pushing high-energy charges into the battery, before stopping to measure the internal impedance of the battery. The computer then decides whether to give another boost, and so on until the battery has been fully charged. The computer then changes to a holding or trickle charge, perhaps after monitoring the self-discharge rate of the battery bank.
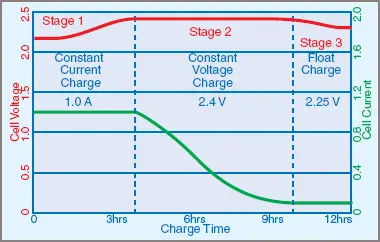
Figure 1 Lead Acid Battery Charging States
Some battery chargers use special charging techniques to ‘recondition’ a battery, removing sulphation, for example, from the inside of the battery.
Battery Management Systems
Larger and more expensive battery banks commonly have battery management systems (BMS), which can be quite complex, using a computerized circuit attached to each battery to monitor voltage and temperature and to adjust the level of charge received by that battery so the individual batteries do not overcharge or overheat.
Electric vehicles using lithium batteries, which can be light but expensive, will typically use a BMS to make the most out of the storage available to the driver.
Battery Maintenance Checklist Key Takeaways
Following proper battery maintenance practices and utilizing advanced charging technologies are vital for ensuring the reliability, efficiency, and longevity of batteries across various applications. From critical backup systems to electric vehicles, consistent upkeep and intelligent management not only enhance performance but also reduce operational risks and costs, making them essential in both everyday and high-demand environments.