This guide covers electrochemistry multiple choice questions (mcqs) with answers and explanation.
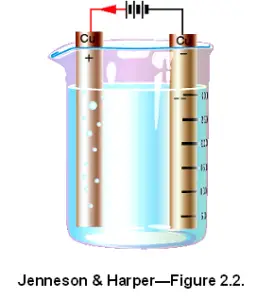
| 1. | In the electroplating process an external voltage is applied across a pair of electrodes, causing:
|
| 2. | In the electroplating process the electrode connected to the positive polarity of the supply is called the anode and the electrode connected to the negative is:
|
| 3. | Primary cells are electrochemical devices that convert chemical energy into:
|
| 4. | Secondary cell chemistries permit the cell to be recharged by:
|
| 5. | Look at the following figure:  The diagram shows a simple voltaic cell consisting of two electrodes, one of copper and the other of zinc, immersed in a solution of dilute hydrochloric acid. In this cell the copper electrode is:
|
| 6. | This simple cell is not very practical, because the copper electrode becomes covered with hydrogen gas, preventing hydrogen ions from taking further electrons from the surface. This effect is known as:
|
| 7. | To reduce the local action in a simple cell it is necessary to:
|
| 8. | Two dissimilar metals in an electrolyte form a primary cell. The voltage across their terminals depends on the:
|
| 9. | Look at the following table: 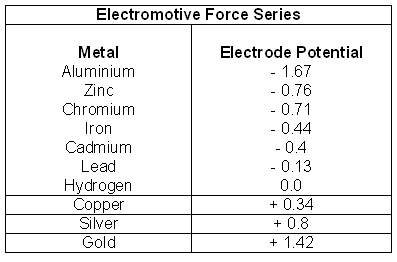 A simple voltaic cell has electrodes of copper and aluminum. The EMF of the cell will be:
|
| 10. | If a small amount of an acid, alkali or salt is added to water, it can become:
|
| 11. | If an electric current flows through the electrolyte, it dissociates the molecules of the substances in solution into:
|
| 13. | 13.The following equation shows the shows the action if copper sulphate is dissolved in water and a current passed through it.
|
| 14. | When an electric current is passed through an electrolyte:
|
| 15. | When the electrolysis method for transferring metal from one electrode to another is used on other metal components to place a metal coating on the surface of that metal, the b process is called:
|
| 16. | Look at the following diagram: 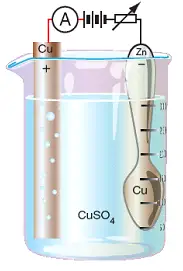 The drawing shows the basic requirements of an electroplating bath. In the drawing, the:
|
| 17. | Anodizing creates an oxide layer on the surface of a metal that is:
|
| 18. | The Electroerosion process can be used to:
|
| 19. | Faraday’s first law of electrochemical deposition states that the mass of metal deposited is directly proportional to the:
|
| 20. | Faraday’s second law of electrochemical deposition states that the mass of metal deposited by a quantity of electric charge is:
|
| 21. | The following formula can be used to determine the amount of metal deposited by an electroplating bath. In the formula, ‘z’ stands for the:
|
| 22. | Look at the following table: 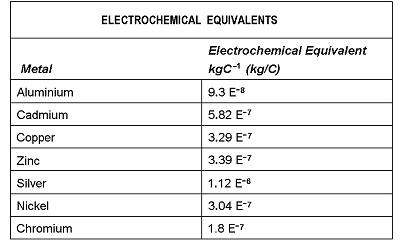 An electrolytic refining bath has a constant current of 4500 A. The amount of zinc deposited by this current in 18 hours will be:
|
| 23. | Processes using the electrolytic method of metal deposition:
|
| 24. | Look at the following diagram:  The diagram shows the connection of a copper conductor to an aluminum conductor. The copper and aluminum will act as electrodes when an electrolyte is present from condensation or rain. In this situation, the metal with the higher potential:
|
| 25. | Dissimilar metals in damp ground and adjacent to each other may produce stray currents. These stray currents can cause underground pipes and cables to:
|
| 26. | Electrolytic corrosion of bronze propellers on ships can be minimized by bolting blocks of zinc to the metal hulls adjacent to the propellers. In this situation, the blocks are called:
|
| 27. | When using the Cathodic protection method of electrolytic protection, an external DC voltage is applied between the equipment to be protected and:
|