| 1. | Separators are usually placed between opposing plates in leads acid cells to:
|
| 2. | Gel-type and paste-type electrolytes have become common in lead-acid cells because they:
|
| 4. | Generally speaking, the difference between a primary and secondary cells is that a secondary cell:
|
| 5. | To produce a higher output voltage cells are connected:
|
| 6. | The number of 1.5 volt cells required to be connected in series to give an output of 9 volts is:
|
| 7. | When two or more strings of cells, or batteries of cells, are connected in parallel in order to increase the available current, they are known as:
|
| 8. | Thirty-six, 2 V, lead–acid cells have been connected in banks of batteries to produce a 24 V supply. The number of banks of batteries will be:
|
| 9. | The ‘nominal voltage’ of a cell is the voltage that the cell should produce with:
|
| 10. | The output voltage of a cell changes when a current is drown from the cell, due to the voltage drop across the:
|
| 11. | The internal resistance of a cell will have a voltage drop across it that adds to the nominal voltage during the charging cycle and:
|
| 12. | Look at the following diagram: 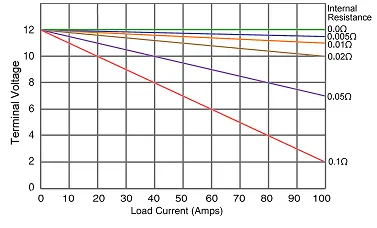 With reference to the terminal voltage-drop chart shown in figure 2, if a load current of 40 A is drawn from a 12 V battery with an internal resistance of 0.05 W, then the terminal voltage will be:
|
| 13. | Generally, the energy that a cell can store depends on the type of cell chemically and the:
|
| 14. | By convention, the energy stored in a cell is measured in:
|
| 15. | The capacity of a lead-acid battery depends on the discharge rate of the battery. A high discharge rate:
|
| 16. | When mixed ready for use in a lead-acid battery, the specific gravity of the diluted sulphuric acid is:
|
| 17. | Look at the following photo:  With reference to figure 3, the picture shows a person carrying out:
|
| 18. | The state of charge of a battery can often be determined from the:
|
| 19. | One type of battery that is rapidly degraded by excessive and repetitive deep discharges is:
|
| 20. | Look at the following table:  With reference to the specific energy table shown in figure 4, the type of cell with the highest energy per kilogram is:
|
| 21. | In automotive terms, the maximum current expected from a battery is called the:
|
| 22. | The efficiency of a battery can be determined using the formula, percentage efficiency =
|
| 23. | The cycle-life of a battery is the number of times they can be:
|
| 24. | Some of the dangers that must not be ignored for when working with cells and batteries are:
|
| 25. | While charging, batteries can vent off a gas such as:
|
| 26. | Lead-acid batteries and alkaline batteries should be kept separated, as the mixing of acids and alkaline can lead to:
|
| 27. | One maintenance procedure with wet cells is to regularly check the electrolyte level because if they are not kept covered they will:
|
| 28. | Cells that are getting old typically have:
|
| 29. | The electrolyte in the zinc-alkaline cell is
|
| 30. | In the zinc-mercury type of cell, the negative electrode is made from:
|
| 31. | When compared with other types, the lithium cell’s output voltage tends to:
|
| 32. | When compared with other types of cell of comparable size and weight, lead-acid cells can deliver:
|
| 33. | As the active materials of a lead-acid cell have no rigid mechanical form or strength they are mounted in a lead grid. The grid usually has antimony added to the lead:
|
| 34. | During the discharge cycle of a lead-acid cell, the sulphuric acid electrolyte ionises in the water to form positive hydrogen ions and:
|
| 35. | Once a lead-acid cell is fully charged, the hydrogen and oxygen, being liberated cannot combine within the cell and:
|
| 36. | For successful operation, the sealed lead-acid cell must be charged at a rate that will:
|
| 37. | One advantage of the lead-acid cell with a gel electrolyte is that they can be used:
|
| 38. | There are two major types of nickel-alkaline cells. One has nickel-iron electrodes and the other has:
|
| 39. | One advantage of the nickel-metal hydride cell secondary cell is that it:
|
| 40. | The modern fuel cell gives more energy per unit mass than a storage battery and in addition:
|
| 41. | The main requirements for batteries for standby power supplies are they must have a long life with a low self-discharge rate and:
|
